 Such standard HIV treatment only works on cells that are actively producing new viral copies. This is in large part because even when suppressed by antiretrovirals, the virus hides in nonreplicating immune cells, known collectively as the viral reservoir. His case has been overseen by a research team led by Asier Sáez-Cirión, head of the viral reservoirs and immune control unit at the Institut Pasteur in Paris. He was treated with radiation, chemotherapy and a stem cell transplant. In 2018, he was diagnosed with a rare blood cancer known as an extramedullary myeloid tumor. He is in his early 50s, was diagnosed with the virus in 1990 and began taking antiretroviral treatment in 2005. The man newly in remission from HIV has been dubbed the Geneva Patient, after the Swiss city where he has received his treatment. Sharon Lewin, president of the IAS and director of the Peter Doherty Institute for Infection and Immunity in Melbourne, Australia, called the new viral remission case “great news.” Such case reports, she said, “help in many ways in the work toward a cure.” The 'Geneva Patient' Scientists generally expect that any success in the effort to develop a widely scalable HIV cure therapy will likely take decades. It remains unethical for a person with HIV who does not already qualify for a stem cell transplant due to cancer to undergo such a treatment in hopes of curing the virus, given such treatment’s considerable toxicity. This major biennial gathering of scientists will also hear noteworthy presentations regarding post-treatment control of HIV in infant boys, circumcision’s impact on HIV risk in gay men, and the relationship between HIV and mpox (formerly known as monkeypox). The ABMF will enable the scale-up of product manufacturing under good manufacturing practice (GMP) guidelines, to support the development of regenerative medicine and cell therapy products used in clinical trials.The man’s case will be presented next week at the International AIDS Society Conference on HIV Science in Brisbane, Australia. In 2018, STEMCELL received a $45 million grant from the Canadian and British Columbian governments toward building an Advanced Biologicals Manufacturing Facility (ABMF) in Burnaby, British Columbia. Since its launch, STEMCELL’s products have expanded to include specialized cell culture and cell separation technologies and services used in stem cell, immunology, cancer, regenerative medicine, and cellular therapy research. as an independent company on July 2, 1993. With a loan from Western Economic Diversification Canada and a mortgage on his house, Eaves purchased the business from the TFL and launched STEMCELL Technologies Inc. Eventually, the demand for MethoCult™ surpassed the capacity of Eaves’ research lab and the TFL. To help fund his blood cancer research program at the TFL, Eaves started selling this medium-named MethoCult™-to other research groups around the world. Allen Eaves’ research group at the TFL made their own standardized cell culture medium for growing hematopoietic stem cells in the lab. 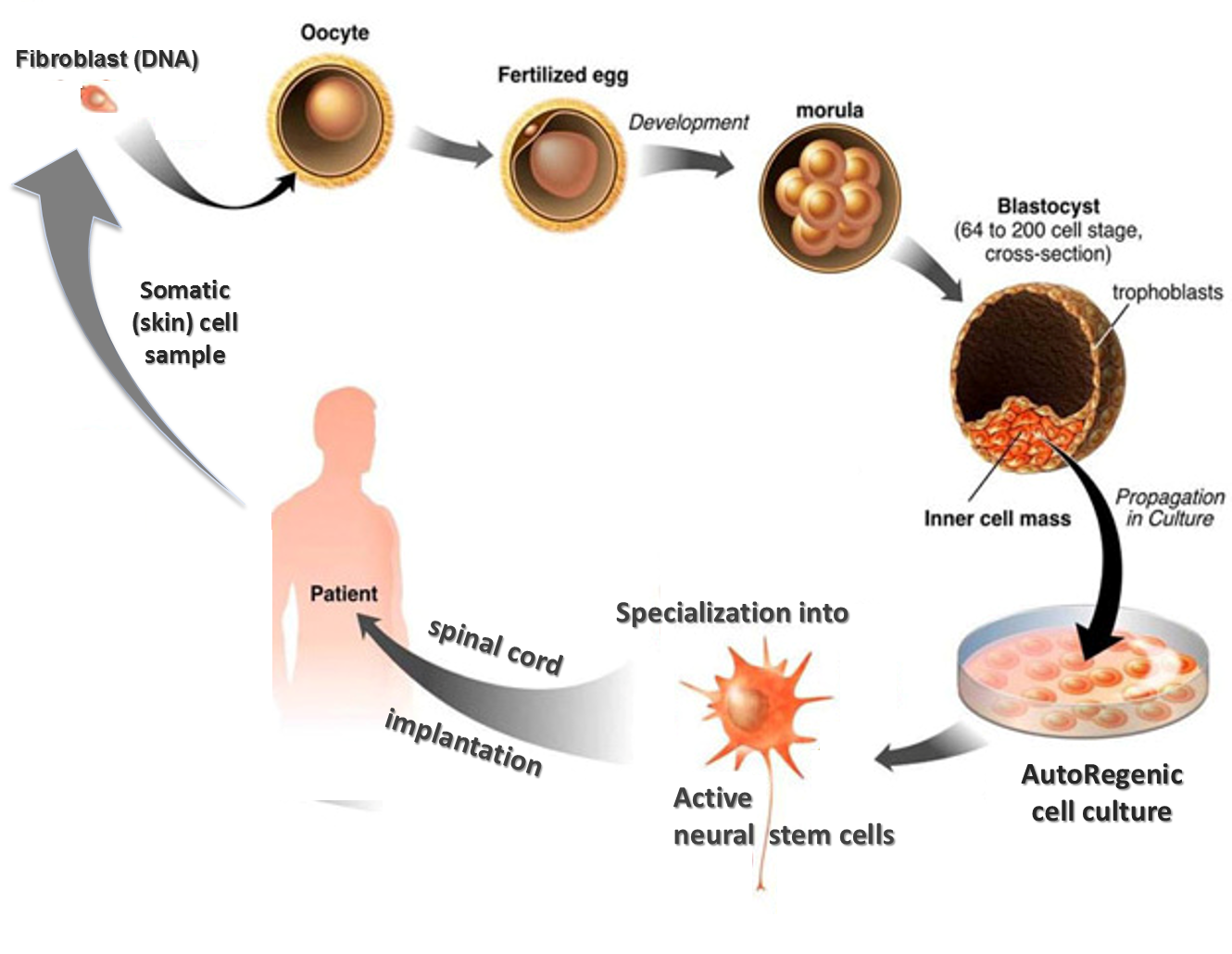
Allen Eaves, the founder, currently serves as the President and CEO.Īllen Eaves (MD, PhD, FRCPC), a hematologist and cancer researcher, co-founded the Terry Fox Laboratory (TFL) in Vancouver with his wife, hematologist and cancer researcher, Dr. The company distributes products to approximately 120 countries.  
The company has offices in the United States, Europe, Asia, and Australia with the headquarter being in Vancouver, British Columbia, Canada. The company also markets education, custom manufacturing, and contract assay services for academic and industrial scientists. STEMCELL Technologies Inc., often abbreviated to STEMCELL, is a Canadian biotechnology company that develops, manufactures, and sells scientific instruments, reagents, and consumables. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
